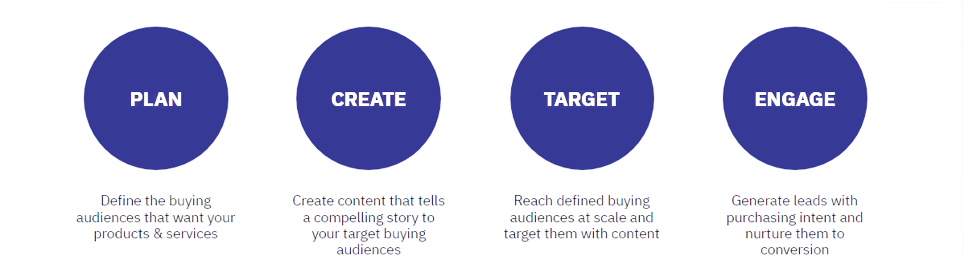
By using insights we draw from GlobalData’s 18 intelligence centers and our powerful companies database that tracks over 500,000 businesses worldwide, we will recommend exactly who you should be targeting and content those targets will be most interested in.
Our automated technology, data analysts and researchers combine to generate over 1 billion data points daily, allowing us to draw on unrivalled real-time insights from all around the world.